
During displacement reactions, a more active nonmetal displaces a less active nonmetal from a compound. Similar to metals, we can also arrange nonmetals in terms of their reactivity. The video shows an experiment to determine the placement of three different metals (Cu, Zn and Mg) on the activity series.See last page which contains an erratum)/Rect/Subj(Notiz)/Subtype/Text/T(cwolke)/Type/Annot>endobj194 0 objendobj195 0 obj/Subtype/Form/Type/XObject>stream Nonmetal activity series is a list in which nonmetals are arranged in the decreasing order of their reactivity. The reactivity series of metals, also known as the activity series, refers to the arrangement of metals in the descending order of their reactivities. If we look at the activity series, we see that aluminum is above copper, so this reaction will occur, and the aluminum will replace the copper to form aluminum sulfate. Metals are found on the left and in the middle, whereas. #"Cu(s) + Zn(NO"_3)_2("aq")"#rarr#"no reaction"#Įxample 2: Will the following single replacement reaction occur? The periodic table can be used to find out if an element is a metal or a non-metal. The oxides of non-metals are acidic or neutral in nature. Oxides of non-metals are formed when it reacts with oxygen. The reactions of the alkali metals in water, exothermic reactions of metals in acid and metal displacement reactions are featured in this video to provide learners with opportunities to explore the reactivity series of metals as part of their 1416 course. The reaction of chlorine with bases like sodium hydroxide gives products like sodium hypochlorite, sodium chloride as well as water. If we look at the activity series above, we see that copper is below zinc, so it cannot replace the zinc in the zinc nitrate compound. The reaction between non-metals and bases is a very complex one. This means that a metal can only replace a metal below it in the series.Įxample 1: Will the following single replacement reaction occur? An activity series of metals lists metals in descending order of reactivity. 
In order to determine whether this will actually happen, you consult an activity series of metals. 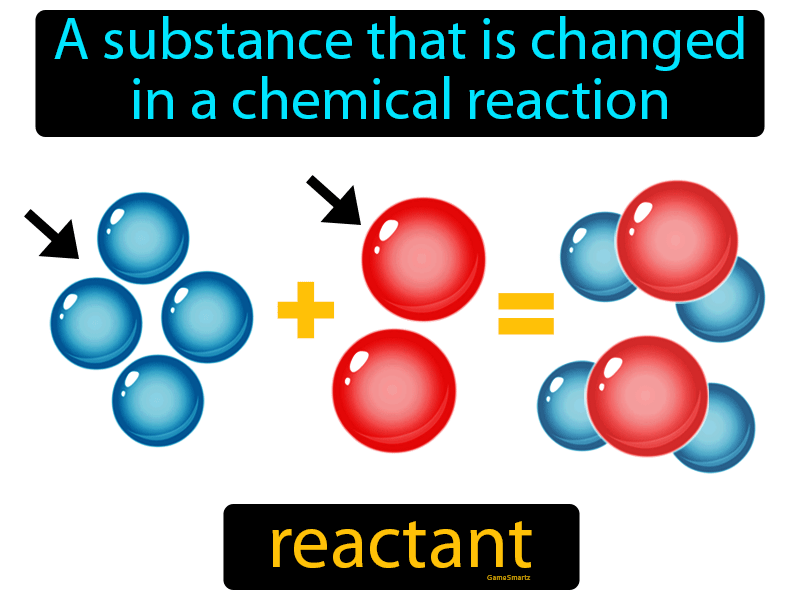
The most reactive elements and compounds may ignite spontaneously or explosively. The reaction can involve the substance on its own or with other atoms or compounds, generally accompanied by a release of energy. Where #"A"# and #"B"# are metals, and #"C"# is a negative ion. In chemistry, reactivity is a measure of how readily a substance undergoes a chemical reaction. As a result of the different ways reactivity can be defined, however, you. 2 3 4 It is used to summarize information about the reactions of metals with acids and water, single displacement. More reactive metals displace less reactive metals from their compounds and react with water. In chemistry, a reactivity series (or activity series) is an empirical, calculated, and structurally analytical progression 1 of a series of metals, arranged by their 'reactivity' from highest to lowest. In a single replacement reaction, a metal and a metal compound may react so that the metal will replace the metal in the compound, producing the replaced metal and a new compound with the original metal. metal will likely react to form copper cations because it is a transition metal. The reactivity series ranks metals by how readily they react.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
